◆Table of contents
ToggleIntroduction
Aluminum is an indispensable material in our lives due to its excellent properties. It has high strength despite being lightweight and is easy to process, so it is used in various fields. However, aluminum is by no means a metal that does not rust. In fact, aluminum can corrode depending on the environment and conditions.
Aluminum has a mechanism to protect itself from corrosion by forming a thin oxide film on its surface by reacting with oxygen. However, this oxide film can be destroyed under certain environmental conditions, and corrosion can progress. Corrosion can not only shorten the life of aluminum products but also affect safety.
Therefore, this article will explain in detail the causes, countermeasures, and specific examples of aluminum corrosion. We will explain in an easy-to-understand manner in what kind of environment aluminum is likely to corrode, what kind of corrosion there is, and how to take measures.
By reading this article, you will be able to deepen your knowledge of aluminum corrosion and take appropriate measures so that you can use aluminum products safely for a longer period of time.
Causes of Aluminum Corrosion
Aluminum is used in various applications due to its excellent properties, but it can corrode under certain environmental conditions. Corrosion can not only shorten the life of aluminum products but also affect safety. Here, we will explain in detail the causes of aluminum corrosion.
Aluminum Corrosion Mechanism
Aluminum Oxide Film and Its Role
When aluminum reacts with oxygen in the air, it forms a thin oxide film on the surface. This oxide film plays a role in protecting aluminum from corrosion. The oxide film is dense and stable, preventing corrosive substances from entering the inside of the aluminum.
Oxide Film Destruction and Corrosion Occurrence Mechanism
However, under certain environmental conditions, this oxide film is destroyed and corrosion progresses. The oxide film is vulnerable to acidic and alkaline environments, environments containing chloride ions, and hot and humid environments, and can be destroyed when exposed to these environments.
When the oxide film is destroyed, the aluminum base material comes into direct contact with corrosive substances, and corrosion begins. Corrosion progresses by electrochemical reaction, and gradually spreads to the inside as aluminum ions dissolve.
Corrosion Progression Mechanism (Chemical Reaction Formula, etc.)
Aluminum corrosion progresses by the following electrochemical reactions.
- Anode Reaction: Al → Al³⁺ + 3e⁻
- Cathode Reaction: O₂ + 2H₂O + 4e⁻ → 4OH⁻
By these reactions, aluminum dissolves into ions and combines with hydroxide ions to produce aluminum hydroxide. Aluminum hydroxide is further oxidized to become alumina hydroxide and accumulates as a corrosion product.
Types of Aluminum Corrosion
There are various types of aluminum corrosion. The following shows the representative types of corrosion and their characteristics.
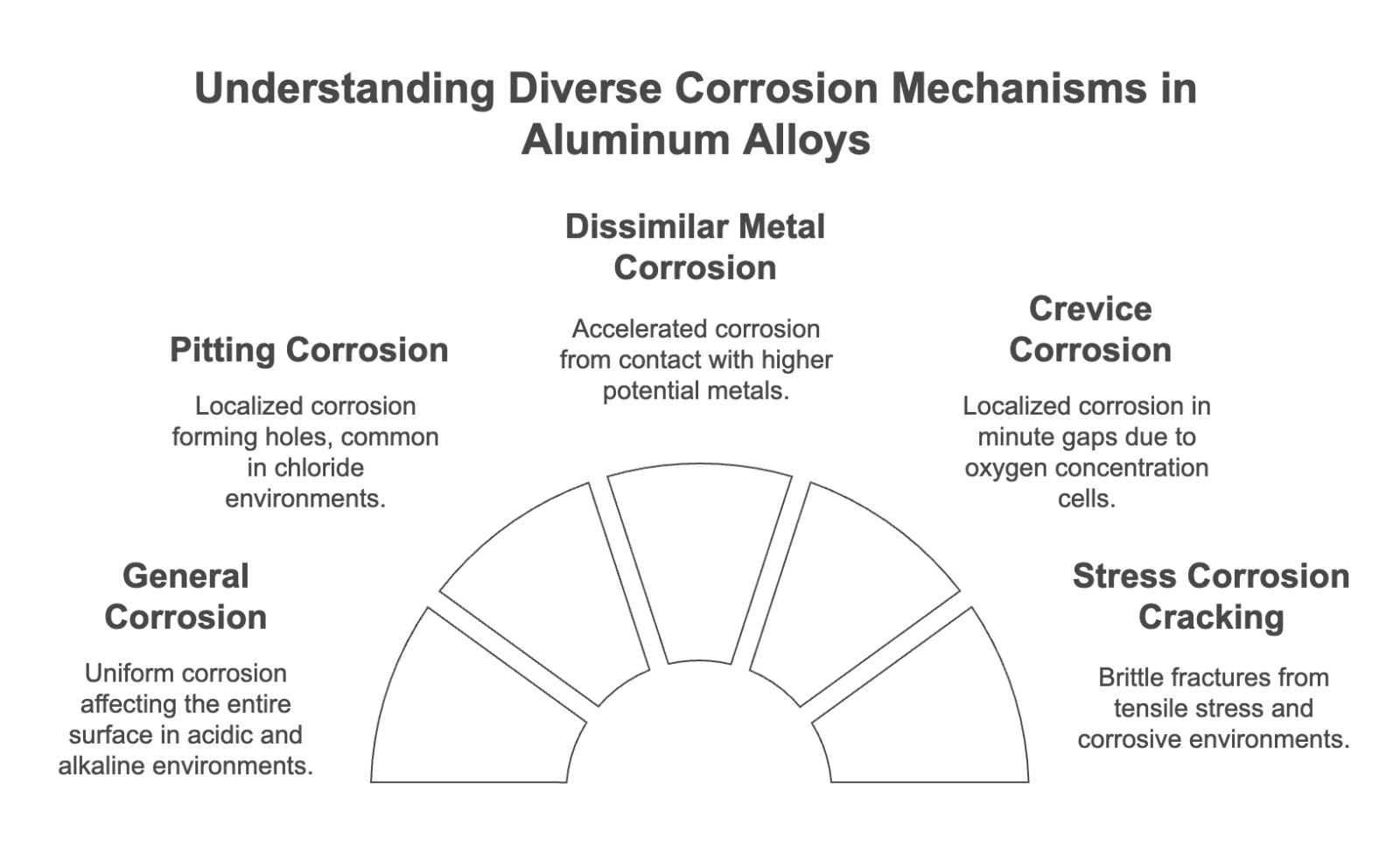
- General Corrosion (Uniform Corrosion): The entire oxide film dissolves uniformly, and the entire aluminum surface corrodes. It tends to occur in acidic and alkaline environments.
- Pitting Corrosion: Localized corrosion concentrates and progresses in holes. It tends to occur in environments containing chloride ions.
- Dissimilar Metal Contact Corrosion: When aluminum comes into contact with a metal with a higher potential than it, aluminum corrosion is accelerated.
- Crevice Corrosion: Oxygen concentration cells are formed in minute gaps, and localized corrosion occurs.
- Intergranular Corrosion: Crystal grain boundaries are preferentially corroded.
- Stress Corrosion Cracking (SCC): Brittle fracture occurs when tensile stress and a specific corrosive environment overlap.
- Others: Filiform corrosion, etc.
Factors Affecting Corrosion
Aluminum corrosion is affected by various factors. The following shows the main factors and their effects.
- Environmental Factors: Humidity, temperature, and pollutants (sulfur dioxide, etc.) promote corrosion.
- pH: In acidic or alkaline environments, the corrosion rate of aluminum increases.
- Contact with Dissimilar Metals: Contact with metals nobler than aluminum accelerates aluminum corrosion.
- Others: Stress, shape, and surface condition can also affect corrosion.
Aluminum corrosion occurs due to the combined effect of various factors. It is important to comprehensively consider these factors when taking corrosion countermeasures.
Aluminum Corrosion Countermeasures
Aluminum corrosion is caused by various factors. To prevent corrosion and use aluminum products for a long time, it is important to take appropriate measures. Here, we will explain the main countermeasures for aluminum corrosion.
| Countermeasure Item | Content | Details |
|---|---|---|
| Material Selection | Selection of highly corrosion-resistant aluminum alloy | Select an appropriate alloy according to the usage environment and application with reference to JIS standards, etc. |
| Surface Treatment | Anodizing | Forms an oxide film to improve corrosion resistance. The effects and precautions vary depending on the type. |
| Painting | Covers the surface with a coating film and blocks contact with corrosive substances. The effect and durability vary depending on the type of paint and the painting method. | |
| Plating | Covers the surface with a thin metal film to impart corrosion resistance. The effect and properties vary depending on the type of plating. | |
| Others | Anodic oxidation treatment, chemical conversion treatment, etc. | |
| Environmental Control | Removal of corrosive substances | Removes corrosive substances by cleaning or filtering. |
| Humidity control | Properly control humidity with dehumidifiers and desiccants. | |
| Temperature control | Properly control temperature with temperature control devices, etc. | |
| Design/Structure | Avoidance of easily corroded shapes | Avoid shapes and gaps where water tends to accumulate. |
| Installation of drain holes | Allow drainage when water enters the inside of the structure. | |
| Prevention of dissimilar metal contact | Prevent contact with dissimilar metals with insulating materials and anti-corrosion paints. | |
| Others | Use of corrosion inhibitors | Use chemicals that inhibit corrosion. |
| Regular maintenance | Perform regular inspections, cleaning, and repairs. |
Material Selection
How to Choose Highly Corrosion-Resistant Aluminum Alloys
There are various types of aluminum alloys, each with different properties. Choosing a highly corrosion-resistant alloy is the basis of corrosion protection.
Alloy Types and Properties (JIS Standards, etc.)
JIS standards specify the types and properties of aluminum alloys. When choosing an alloy, refer to JIS standards and choose an appropriate alloy according to the usage environment and application.
Points for Alloy Selection According to Application
For example, when using in a marine environment, it is necessary to choose an alloy with high seawater resistance. Also, when using in a high-temperature environment, it is necessary to choose an alloy with high heat resistance.
Surface Treatment
Anodizing: Principle, Types, Effects, Precautions
Anodizing is a treatment that forms an oxide film on the aluminum surface to improve corrosion resistance. There are various types of anodizing, each with different effects and precautions.
Painting: Types, Effects, Painting Methods, Precautions
Painting is a treatment that covers the aluminum surface with a coating film and blocks contact with corrosive substances. The effect and durability vary depending on the type of paint and the painting method.
Plating: Types, Effects, Plating Methods, Precautions
Plating is a treatment that forms a thin metal film on the aluminum surface to impart corrosion resistance. The effect and properties vary depending on the type of plating.
Other Surface Treatments: Anodic Oxidation Treatment, Chemical Conversion Treatment, etc.
In addition to anodizing, painting, and plating, there are various surface treatments such as anodic oxidation treatment and chemical conversion treatment. These treatments have different effects and applications.
Environmental Control
Removal of Corrosive Substances: Cleaning Methods, Filters, etc.
Corrosive substances (chloride ions, acidic substances, etc.) promote aluminum corrosion. It is important to remove corrosive substances by regular cleaning and the use of filters.
Humidity Control: Dehumidifiers, Desiccants, etc.
High humidity environments promote aluminum corrosion. It is important to properly control humidity using dehumidifiers and desiccants.
Temperature Control: Temperature Control Devices, etc.
High temperature environments promote aluminum corrosion. It is important to properly control temperature using temperature control devices.
Design/Structure
Avoidance of Easily Corroded Shapes: Water Puddles, Gaps, Stress Concentration
Shapes where water tends to accumulate and structures with many gaps are prone to corrosion. It is important to avoid these shapes and structures from the design stage.
Installation of Drain Holes: Position, Shape, Precautions
It is important to install drain holes so that water can be drained quickly when it enters the inside of the structure. Install drain holes considering their position, shape, and precautions.
Prevention of Dissimilar Metal Contact: Insulating Materials, Anti-Corrosion Paints, etc.
When aluminum comes into contact with a metal with a higher potential than it, corrosion is accelerated. It is important to prevent contact with dissimilar metals using insulating materials and anti-corrosion paints.
Others
Use of Corrosion Inhibitors: Types, Effects, Precautions
Corrosion inhibitors are chemicals that inhibit corrosion. There are various types of corrosion inhibitors, each with different effects and precautions.
Regular Maintenance: Inspection Items, Cleaning Methods, Repair Methods
Regular maintenance is important to detect and deal with corrosion of aluminum products early. Properly define inspection items, cleaning methods, repair methods, etc., and carry out maintenance regularly.
Aluminum Corrosion Cases and Countermeasures
Aluminum corrosion occurs in various environments and conditions. Here, we will explain typical corrosion cases and their countermeasures.
Dissimilar Metal Contact Corrosion Cases
Corrosion Due to Contact Between Stainless Steel and Aluminum
Stainless steel and aluminum are a combination that easily causes dissimilar metal contact corrosion. For example, if an aluminum member is fixed with stainless steel bolts, the aluminum may corrode.
Identification of Corrosion Causes and Countermeasures
This corrosion occurs due to the potential difference between stainless steel and aluminum. Since stainless steel has a higher potential than aluminum, aluminum becomes a sacrificial anode and corrosion is accelerated.
Countermeasures
- Materials: Make contact with the same metal, such as using aluminum bolts.
- Design: Insert an insulating material (rubber, plastic, etc.) between stainless steel and aluminum.
- Surface Treatment: Apply anti-corrosion paint to the aluminum side.
Pitting Corrosion Cases
Pitting Corrosion in Environments Containing Chloride Ions
In environments containing chloride ions (coastal areas, snow melting agent spraying areas, etc.), pitting corrosion is likely to occur in aluminum. Pitting corrosion is a phenomenon in which small holes open on the aluminum surface and corrosion progresses inward.
Identification of Corrosion Causes and Countermeasures
Pitting corrosion occurs when chloride ions destroy the oxide film and induce corrosion.
Countermeasures
- Materials: Use aluminum alloys with high pitting corrosion resistance (A5052, etc.).
- Environmental Control: Wash with water regularly to remove chloride ions adhering to the surface.
- Surface Treatment: Apply anodizing or painting to strengthen the oxide film.
Other Corrosion Cases
Corrosion Cases in Specific Products and Environments
- Cases where pitting corrosion and dissimilar metal contact corrosion occurred on aluminum outer plates of ships used in seawater environments.
- Cases where corrosion due to acidic water occurred in aluminum pipes used in hot spring areas.
- Cases where corrosion due to snow melting agents occurred on aluminum wheels of automobiles.
Identification of Corrosion Causes and Countermeasures
In these cases, it is important to identify the causes of corrosion and take appropriate measures. For example, if it is a seawater environment, it is necessary to select an alloy with high seawater resistance, and if it is an acidic water environment, it is necessary to select an alloy with high acid resistance. It is also important to conduct regular maintenance and inspections to detect and counter corrosion early.
Standards and Criteria for Aluminum Corrosion
It is very important to understand the appropriate standards and criteria when taking measures against aluminum corrosion. Here, we will explain the representative standards and criteria.
JIS (Japanese Industrial Standards)
JIS (Japanese Industrial Standards) is a standard established based on the Industrial Standardization Act of Japan. JIS standards for aluminum and aluminum alloys include the following.
- JIS H 4000: Aluminum and aluminum alloy sheets and strips
- JIS H 4040: Aluminum and aluminum alloy extruded shapes
- JIS H 8601: Anodic oxide coatings on aluminum and aluminum alloys
- JIS H 8681: Method of corrosion resistance test for anodic oxide coatings on aluminum and aluminum alloys
These standards define the quality and performance of aluminum materials, test methods, etc., and play an important role in product design, manufacturing, and evaluation.
ASTM (American Society for Testing and Materials) Standards
ASTM (American Society for Testing and Materials) is an American material standards organization. ASTM standards are widely adopted around the world, and there are many standards for aluminum and aluminum alloys.
- ASTM B209: Aluminum and Aluminum-Alloy Sheet and Plate
- ASTM B211: Aluminum and Aluminum-Alloy Rolled or Extruded Bar, Rod, and Wire
- ASTM B221: Aluminum and Aluminum-Alloy Extruded Structural Shapes
- ASTM B244: Standard Test Method for Measuring Thickness of Anodic Coatings on Aluminum and Other Anodic Coatings
These standards define the properties and test methods of aluminum materials, and play an important role in international transactions and product development.
Other Related Standards
In addition to the above, there are many standards and criteria related to aluminum corrosion.
- ISO (International Organization for Standardization) Standards: International standards, and there are many standards for aluminum.
- Industry Association Standards: Standards independently established by each industry association, and the content is tailored to the needs of each industry.
These standards and criteria play an important role in ensuring the quality of aluminum products and properly implementing corrosion countermeasures. In product design, manufacturing, and evaluation, refer to the relevant standards and criteria and take appropriate material selection and corrosion countermeasures.
Latest Information on Aluminum Corrosion
Corrosion protection for aluminum is constantly evolving. Here, we will introduce the latest research trends and technical information.
Latest Research Trends
Development of High-Strength Alloys
In order to meet the needs for weight reduction of aircraft and automobiles, the development of high-strength aluminum alloys is progressing. These alloys have higher strength than conventional alloys, while also having excellent formability and weldability.
Development of Heat-Resistant Alloys
High heat resistance is required for aluminum products used in high-temperature environments such as engine parts. Therefore, the development of new alloys with excellent heat resistance is also progressing. These alloys maintain high strength even at high temperatures and improve product reliability.
Development of Environmentally Friendly Alloys
With the growing awareness of environmental issues, the development of aluminum alloys with low environmental impact is also attracting attention. Alloys that are easy to recycle and alloys that can reduce energy consumption during manufacturing are being developed, contributing to the realization of a sustainable society.
New Corrosion Protection Technologies
Self-Healing Coatings
In recent years, coating technologies with self-healing functions have been developed. This coating automatically repairs itself even if the surface is scratched, so it can suppress the progress of corrosion.
Surface Treatment Utilizing Nanotechnology
New surface treatment technologies utilizing nanotechnology have also appeared. These technologies can dramatically improve corrosion resistance by forming a special film on the aluminum surface.
Others
- Corrosion prediction technology utilizing AI (artificial intelligence)
- Corrosion monitoring technology using IoT (Internet of Things) sensors
These latest technologies are expected to greatly contribute to extending the life of aluminum products.
Summary
Aluminum is a convenient metal that is lightweight and easy to process, but it can corrode under certain environmental conditions. Corrosion not only shortens the product life but also affects safety, so appropriate measures are important.
Aluminum corrosion occurs when the oxide film on the surface is destroyed. Causes include acidic and alkaline environments, chloride ions, high temperature and humidity. Corrosion includes general corrosion, pitting corrosion, dissimilar metal contact corrosion, etc.
Countermeasures include material selection, surface treatment, environmental control, design/structure, and others. In material selection, choose a highly corrosion-resistant alloy, and in surface treatment, perform anodizing, painting, plating, etc. In environmental control, remove corrosive substances and control humidity and temperature, and in design/structure, avoid easily corroded shapes and install drain holes. Others include the use of corrosion inhibitors and regular maintenance.
Aluminum corrosion occurs due to the combined effect of various factors. It is important to comprehensively consider these factors in countermeasures. Conduct regular maintenance and strive for early detection and countermeasures of corrosion.
